Chondrosarcoma is a malignant neoplasm in which tumor cells produce various amounts of neoplastic chondroid and fibrillary matrix, but not osteoid matrix [
1]. This is the second most common primary tumor of bone in humans and dogs and accounts for approximately 5% to 10% of all canine primary bone tumors [
2].
In dogs, chondrosarcoma occurs most often in medium to large breeds, particularly boxers, German shepherds, and golden retrievers, but is rare in small and giant breeds [
1,
3]. Moreover, the tumor is most common in middle-aged to older dogs; the mean age of the affected animals ranges from 5.9 to 8.7 years [
3].
Mesenchymal chondrosarcoma is a rare subtype of chondrosarcoma among the World Health Organization classifications and accounts for approximately 1% of all chondrosarcomas in humans [
4]. Histologically, mesenchymal chondrosarcomas are characterized by a transition from undifferentiated mesenchymal cells and an array of variously differentiated chondroid components [
1]. On the other hand, chondrosarcoma is relatively rare in most domestic species. This paper presents a rare case of mesenchymal chondrosarcoma in the maxillary gingiva of a small-breed Maltese dog.
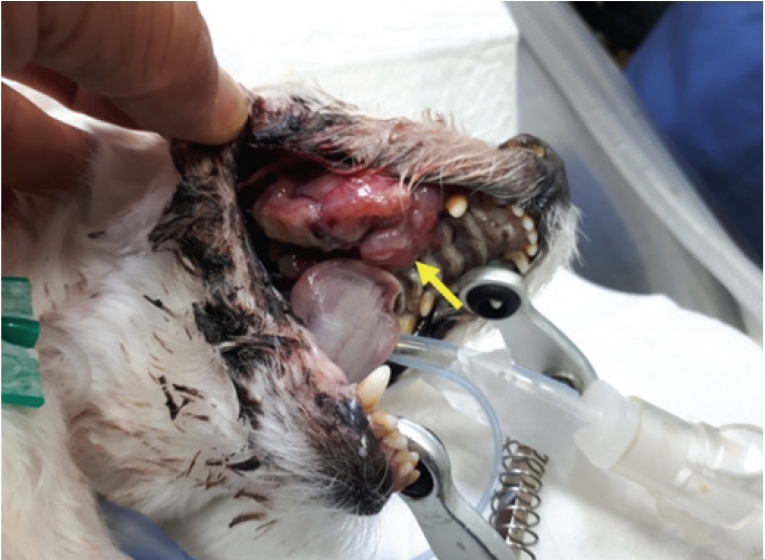
A 13-year-old castrated male Maltese dog was presented to a local animal hospital with an oral hemorrhage and swelling on the right side of his face. An intraoral examination revealed an irregular proliferated lobular mass at the entire right side of the gingival region and hard palate (
Fig. 1). A radiography examination showed a soft tissue mass lesion in the right maxillary region without metastases to other organs. Blood analysis revealed mild anemia (hematocrit, 29.8%; reference range, 35%-56%) and mild leukocytosis (21.8 L/┬ĄL; reference range, 5.0-20.0 L/┬ĄL). The serum biochemical results revealed moderate elevations in the alkaline phosphatase (113 U/L; reference range, 17-78 U/L).
The dog did not respond after a 1-week treatment with steroids, and the edematous lesion worsened. The edema resulted in the caudal and dorsal displacement of the right eyeball. An incisional biopsy sample was presented to the Pathology Department of the Veterinary Medicine, Jeju National University, for a histopathological examination. The requested mass was approximately 3.5 ├Ś 1.8 ├Ś 1.8 cm in size. The cut surface was firm and tan to white.
The mass was fixed in 10% neutral-buffered formalin, trimmed and processed according to standard protocols, and embedded in paraffin wax. The paraffin sections (4 ╬╝m) were stained with hematoxylin and eosin and Alcian blue (pH 1.0 and 2.5) for the microscopic examination. To determine the origin of the tumor cells, replicated paraffin sections of the mass were also subjected to immunohistochemical analysis and incubated with the following primary antibodies: anti-vimentin (mouse monoclonal antibody, 1:100; Dako, Denmark) and anti-S-100 (rabbit polyclonal antibody, 1:400; Dako).
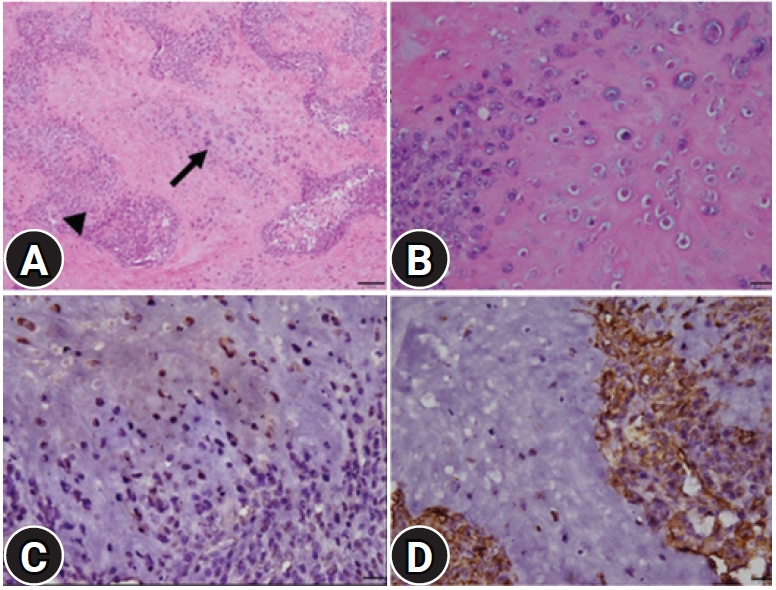
The microscopy examination showed that the neoplastic foci comprised a biphasic morphological pattern, such as primitive mesenchymal tissue and mature or immature cartilage tissues (
Fig. 2A). The primitive mesenchyme was composed of round and oval or mild elongated immature neoplastic cells with high cellularity. These cells had scant pale eosinophilic cytoplasm and indistinct cellular borders with some mitotic figures. The cartilage areas were scattered throughout the lesions. The tumor cells were arranged irregularly within the lacunae of a pale, basophilic or eosinophilic, intercellular matrix (
Fig. 2B). These neoplastic cells had abundant pale-staining cytoplasm, moderate anisokaryosis, and round vesicular nuclei with prominent nucleoli. Mitotic figures were rare. A small number of multinucleated giant cells were present within the mass. Severe multifocal necrosis was also present around the mass, particularly in the peripheral area. The cartilaginous materials and lacunae stained greenish-blue, and the nuclei of chondrocytes stained red in contrast to the Alcian blue stain. Through immunohistochemical analysis, most neoplastic cells forming multifocal cartilaginous islands were strongly positive for the S-100 protein (
Fig. 2C); the surrounding mesenchymal cells were strongly positive for vimentin (
Fig. 2D).
Chondrosarcomas usually occur in older dogs from large breeds, and there is no sex predilection. To the best of the authorsŌĆÖ knowledge, this is the first report of mesenchymal chondrosarcoma occurring in the maxillary gingiva of a small-breed Maltese dog.
In dogs, as in humans, chondrosarcomas more often involve the flat bones than the long bones. In a retrospective study of 97 dogs with chondrosarcomas, the nasal cavity was the most common site (28.8%), followed by the ribs (17.5%), appendicular skeleton (17.5%), extraskeletal sites (13%), and facial bones (9%) [
3]. In another study, 31 out of 35 cases (88.6 %) involved the flat bones, predominantly the ribs, turbinates, or pelvis [
2].
Extraskeletal chondrosarcomas are rare soft tissue sarcomas arising from soft tissues and visceral organs [
5]. In dogs, approximately 1% of chondrosarcomas occur in extraskeletal locations, including the lungs, retroperitoneum, omentum, and spleen [
6-
9].
Histologically, two types of chondrosarcoma are recognized: conventional, the most common type found in skeletal tumors, and mesenchymal, a rarer type found more often in the extraskeletal sites [
10]. Myxoid or conventional chondrosarcoma consists of spindle-shaped cells trapped within a basophilic hyaline matrix. Mesenchymal chondrosarcoma has been described as having two distinct histologic patterns. The first is moderate to well-differentiated chondroid tissue, while the second is described as undifferentiated malignant mesenchymal cells arranged in sheets [
1]. The histological appearance of the oral mass, in this case, was similar to that described for mesenchymal chondrosarcoma in humans and dogs.
The diagnosis of biopsy specimens remains a challenge because of the variable tumor components, and specimens may contain only one of the two elements. The absence of cartilage in the biopsy sample can confuse this tumor with other small, round, blue-cell tumors. The histological differential diagnosis of mesenchymal chondrosarcoma included lymphoma, neuroblastoma, small cell osteosarcoma, and embryonal rhabdomyosarcoma [
11]. Large samples of healthy tissue at the edge and center of the lesion will increase the diagnostic yield. Moreover, typical immunohistochemistry findings include the positivity of the mesenchymal portion for the vimentin and the positivity of the cartilaginous regions for the S-100 protein. In this case, the requested mass was relatively large and contained two elements essential for diagnosis. Moreover, the mass showed similar immunohistochemical findings to those previously reported [
12].
In a diagnostic examination for chondrosarcomas, radiographs only provide the anatomical location of the tumor [
13]. Computed tomography can evaluate the degree of bone invasion, and magnetic resonance imaging can reflect the increased aqueous contents of abundant neoplastic cartilage matrix in a T2-weighted hyperintense area, particularly in well-differentiated low-grade chondrosarcoma, as reported in humans [
13]. The dog, in this case, was examined using only radiographs, so no evidence of bony involvement was observed within the lesion.
Extraskeletal chondrosarcomas occurring in the oral cavity are exceedingly rare. In a previous study, among 109 malignant oral neoplasms in dogs, none were classified as chondrosarcoma [
14]. Extraskeletal chondrosarcoma of the gingiva has been reported in rabbits and humans [
15]. In rabbit chondrosarcoma, the tumor invaded the nasal cavity and the survival time after diagnosis was 15 days [
15].
In the present case, the dog only had a portion of the mass removed for biopsy; no other aggressive treatment, such as radical surgery with a wide margin or systemic chemotherapy, was performed. After diagnosis, the dog was treated only with antibiotics, and the remaining tumor recurred more significantly. The prognosis was poor in this case, and the dog died two months after diagnosis. Although mesenchymal chondrosarcoma of the maxillary gingiva is rare, clinicians and pathologists should be aware of this rare entity when making a differential diagnosis of oral tumors.









 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



