In vivo and in vitro efficacy of florfenicol, terbinafine, and mometasone furoate topical otic solution for the treatment of canine otitis externa
Article information
Abstract
This study evaluated the efficacy of a new, single-dose otic solution combining florfenicol, terbinafine, and mometasone furoate for the treatment of canine otitis externa (OE) in vitro and in vivo. Forty-one client-owned dogs with OE were included in the study and divided into a treatment group that received the test solution and a negative control group that received a normal saline solution. On day 0, the dogs were treated either with the test or the control solution and evaluated over 30 days. Clinical efficacy was evaluated by clinical signs and cytological organism counts. In vitro antimicrobial susceptibility was evaluated by the minimum inhibitory concentration (MIC). After treatment with the test solution, clinical signs continuously decreased and cytological scores were significantly reduced. The results of MIC testing showed that the test solution was potent against the common pathogenic causes of canine OE. In this study, the most common causative pathogens were Staphylococcus pseudintermedius, Pseudomonas spp. and Malassezia pachydermatis. No issues related to safety were identified. Based on these results, this new ototopical drug can be used as first line treatment for canine OE.
Introduction
Canine otitis externa (OE) is one of the most common dermatological problems for which dog owners frequently seek veterinary care and is associated with a high rate of recurrence [1-3]. Dogs with OE may show erythema, swelling, exudate, and clinical signs related to pruritus and pain due to inflammation in the external ear canal [3,4].
Numerous underlying issues can cause or worsen OE; however, addressing pathogens such as bacteria or yeast plays an important role in the management of OE. Although various pathogens are associated with canine OE, Staphylococcus pseudintermedius, Pseudomonas aeruginosa, and Malassezia pachydermatis are the most common pathogens found in this condition [3-7]. Thus, reducing number of these infectious organisms is critical to the successful treatment of OE. For this purpose, topical and systemic antimicrobials with or without anti-inflammatory agents are used in veterinary clinics [3].
The otic solution used in this study (Neptra; Elanco, USA) includes florfenicol, terbinafine, and mometasone furoate in a single-dose regimen. This otic solution is the Food and Drug Administration−approved, veterinarian−administered treatment for canine OE, and S. pseudintermedius and M. pachydermatis show susceptibility to the active ingredient. Similar to chloramphenicol, florfenicol is a broad-spectrum antibiotic with efficacy against many bacteria. This drug was investigated for use in treating respiratory diseases in domestic animals as well as dermatologic diseases in small animals [8-10]. Terbinafine is an antifungal agent used primarily for dermatophytosis but may be useful for Malassezia spp. dermatitis [11-13]. Finally, mometasone furoate is a highly potent topical corticosteroid used to treat pruritic and/or inflammatory conditions in skin infection [14]. A previous study demonstrated the efficacy of this formulation in the treatment of OE [15]. In addition, the active ingredients in this solution are not generally used in Korea, thus we explored the merits of this otic solution on canine OE in our country.
The purpose of this study was to evaluate the in vivo and in vitro efficacy of an otic solution containing florfenicol, terbinafine, and mometasone furoate for the treatment of canine OE.
Materials and Methods
Animals
Dogs diagnosed with OE caused by bacterial and fungal infection with any clinical signs were included. Inclusion criteria further included a minimum age of 12 weeks and intact tympanic membrane. Animals with any concurrent ear disease associated with OE (e.g., neoplasia, polyp, or parasite otitis) or with any systemic disease requiring treatment, such as immune-mediated or hormonal disease, were excluded. Dogs administered with anti-infective, and antifungal and anti-inflammatory agents during the last 4 weeks prior to the study were also excluded.
Study design
Before the study, the experiment process was briefed to the dog’s owners, and they consented to their dogs’ inclusion in the study. The animals were randomly divided into the group that received the test solution or the other group that received a saline solution as negative control. Prior to the application of each solution, ear cleaning was performed using warm saline solution. The dosage for both, the test and control solution, was 1 mL per one ear.
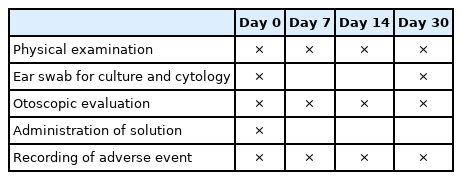
The animals were observed on day 0, day 7 ± 2, day 14 ± 2, and day 30 ± 2. The overall schedule of events is shown in Table 1. Throughout the study clinical examination and scoring were performed using otoscopic examination and total clinical scoring (TCS) system. The TCS system evaluated for erythema, swelling, erosion/ulceration, and exudate based on severity scores ranging from 0 (absent) to 3 (severe). The primary endpoint of the efficacy evaluation was expressed as success or failure of the treatment. If the TCS score was less than 3 points on the last visit day and the scores for each evaluation factor of TCS did not deteriorate, the treatment was judged as a “success”.
At the initial and final visits, ear swabs for pathogen culture and cytology were conducted. Each ear exudate sample was smeared onto a clean slide and stained with Diff-Quik. The clinician scanned the slide and evaluated infectious organisms at ×1,000 magnification (high power field). Five to ten high power fields were scanned in each evaluation, and average counting number of infectious organisms were used for scoring. The scoring criteria for bacteria and yeast count on cytology are shown in Table 2 [16]. The secondary endpoint of the efficacy evaluation was also expressed as “success” or “failure” of the treatment. If the cytology scores per bacteria or Malassezia decreased more than 2 points on day 30 compared with day 0, the treatment was judged as successful.
Microbiological culture and minimum inhibitory concentrations tests
Ear exudate samples were cultured in 5% sheep blood agar and MacConkey agar at 37℃ for 24 hours and Sabouraud dextrose agar at 35℃ for 24 to 48 hours aerobically. The identification of organisms was done by polymerase chain reaction. The minimum inhibitory concentration (MIC) results were interpreted as the lowest concentration of antimicrobial agent that completely inhibited the growth of the organism. The isolated bacteria and yeast specimens were tested for MIC against florfenicol and terbinafine, respectively. The testing was conducted by microdilution as recommended by the Clinical and Laboratory Standards Institute [17,18].
Safety evaluation
At each of the visits, the investigator conducted a physical examination for the evaluation of the safety of the test solution. In addition, the investigator educated the owner to contact or visit an animal hospital in the case of an adverse event such as for example head tilt, nystagmus, or circling during the study period.
Statistical analysis
The experimental unit was the individual dog. In bilateral OE, both ears were treated, but the ear with the higher entry score was selected as the study ear. In the case of equal scores, the right ear was determined to be the study ear.
All analyses were performed with GraphPad Prism version 9.0.0 for Windows (GraphPad Software Inc., USA). The difference in treatment success rates between the 2 groups was evaluated using Fisher exact test at a significance level of 0.05 or less. Quantitative analyses for TCS and cytological evaluation were performed using Wilcoxon code ranking test and Mann-Whitney U-test.
Results
Animals
A total of 41 dogs were included in the study with 28 subjects receiving the test solution and 13 the control solution. Two subjects in the treatment group dropped out because of owner non-compliance. Both subjects did not undergo efficacy evaluation, but MIC was performed from exudate collected from one of them. During the clinical trial period, the subjects were kept in their usual living conditions. The most common breed was the Maltese (26.8%, 11/41), followed by the Poodle (14.6%, 6/41). The mean age was 7 years (range, 1 to 18 years). Animal experiments were performed according to the Guide for the Institutional Animal Care and Use Committee of the Kyungpook National University (IACUC-2019-0117).
Clinical efficacy
TCS evaluation
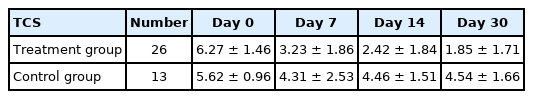
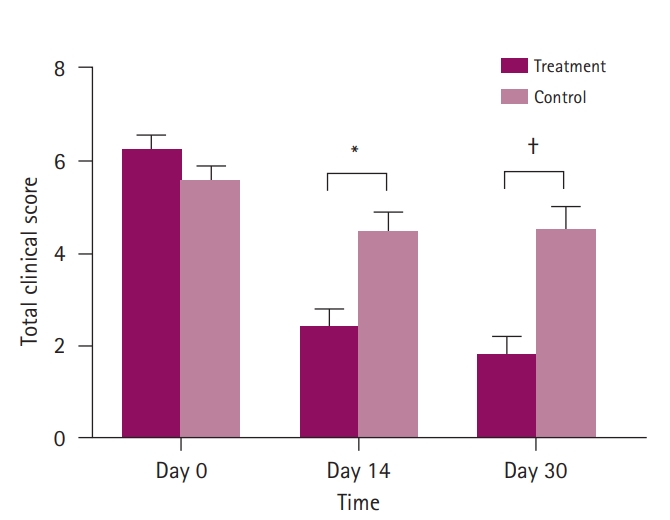
The average TCS scores on day 0, day 7, day 14, and day 30 of each group are shown in Table 3 and Fig. 1. The mean TCS on day 0 of the treatment and control groups were 6.27 and 5.62, respectively and no significant difference in the mean TCS was found between the groups. On day 14, the mean TCS of the treatment group was 2.42, which was lower than that of the control group at 4.46. On day 30, the mean TCS of the treatment group decreased to 1.85; however, that of the control group on day 30 increased to 4.54. The differences in the mean TCS between treatment and control group were statistically significant on day 14 and day 30 (p < 0.01 and p < 0.001, respectively). The treatment success rate based on TCS (primary efficacy endpoint) was 88.5% (23/26) in the treatment group and 7.7% (1/13) in the control group.
Cytological evaluation
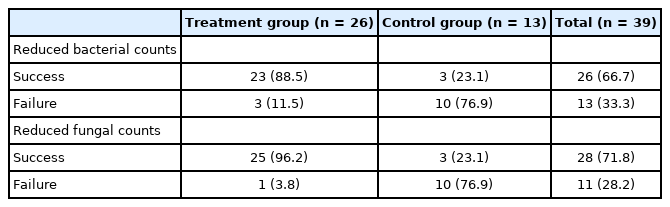
Cytological evaluation using organism counts was conducted on day 0 and day 30. Table 4 shows the success rates of bacterial and yeast cytology score reductions based on the secondary endpoint in the treatment and control groups. A significant reduction in organism count was achieved in the treatment group (p < 0.001).
Secondary endpoint efficacy evaluation findings are shown in Table 4. The success rate of bacterial reduction was 88.5% and 23.1% in the treatment and control group, respectively. The success rate of yeast reduction was 96.2% and 23.1% in the treatment and control groups respectively. These results indicate a statistically significant difference in the reduction of organism count in the treatment group (p < 0.001).
Microbiological culture and MIC tests
A total of 54 bacteria and 50 fungi were isolated, and subsequent MIC evaluation was conducted on the pathogens (Table 5). Florfenicol showed an inhibitory effect on the growth of S. pseudintermedius from concentrations of 4 to 16 μg/mL in vitro. The MIC ranges of P. aeruginosa and Staphylococcus schleiferi were observed at florfenicol concentrations of 2 to 16 μg/mL and 2 to 8 μg/mL, respectively. Terbinafine showed a growth inhibitory effect against M. pachydermatis at a concentration from 0.03 to 16 μg/mL.
Safety evaluation
The animals included in this study were physically examined on each visit day, and owners were asked for any adverse reactions or side effects they had observed since the last visit. No adverse events were observed by the clinician or reported by the owners at any time point during the study.
Discussion
In this clinical study, the test solution was evaluated for safety and therapeutic efficacy in vitro and in vivo.
In vivo clinical efficacy of the otic solution was determined by TCS. On day 0, the TCS of the treatment and control group were 6.27 and 5.62, respectively, and no significant difference was observed between the 2 groups. After treatment, the TCS continuously decreased to 2.42 on day 14 and 1.85 on day 30 in the treatment group, while no relevant improvement of the TCS was observed in the control group (4.46 and 4.54 on day 14 and day 30, respectively). This result shows that the efficacy of the otic solution was maintained throughout 30 days from application.
The treatment success rate based on TCS (primary efficacy endpoint) was 88.5% in the treatment group with 23 out of 26 dogs successfully treated. On the contrary, only one out of 13 dogs was treated successfully in the control group, and overall success rate was 7.7%. TCS consisted of assessments of 4 clinical signs associated with inflammation. Mometasone furoate is a synthetic anti-inflammatory glucocorticoid [14]. The anti-inflammatory effect of mometasone furoate may reduce exudation and swelling and, as a result, increase the effectiveness of the antimicrobial agent. Therefore, this otic solution can effectively manage canine OE symptoms.
In cytologic evaluation, successful decrease in bacterial counts was observed in the treatment group. The reduction rate in the treatment group was considerably higher than in the control group (88.5% versus 23.1%). In terms of yeast count, the treatment group had a reduction rate of 96.2%, whereas the control had 23.1%. Therefore, we considered this otic solution effective in reducing the pathogen count, thereby improving the cytologic score.
The efficacy of the otic solution against pathogenic activity was also demonstrated in vitro. The MIC50 values of florfenicol to suppress bacterial growth were 8 μg/mL, 16 μg/mL, and 8 μg/mL for S. pseudintermedius, P. aeruginosa, and S. schleiferi, respectively. The MIC50 and MIC90 of terbinafine to control Malassezia activity was 0.06 μg/mL and 0.125 μg/mL, respectively. Based on the MIC values obtained, the otic solution seems to be more effective on Staphylococcus spp. than Pseudomonas spp. In a previous study, the florfenicol concentration from an ear flush sample at 10 days after application of the otic solution was 8.7 μg/mL, whereas the terbinafine concentration at this time point was 4.8 μg/mL [15]. These concentrations were higher than the MIC50 values of Staphylococcus spp. and Malassezia spp. in our study. Thus, we presume that one application of the otic solution can sustain efficacy over a few days.
S. pseudintermedius, P. aeruginosa, and M. pachydermatis are the most common causative pathogens in canine OE [3-7]. In the present study, S. pseudintermedius and Pseudomonas spp. were observed in 66.7% (26/39) and 17.9% (7/39) of the subjects, respectively. Malassezia was found in all cases. The most common multiple infections involved S. pseudintermedius and M. pachydermatis, which confirm the previous research findings. Thus, the combined efficacy of florfenicol and terbinafine in this solution may be considered as a first option for veterinarians to treat common dogs with OE.
Antimicrobial resistance affects the successful treatment of canine OE and can lead to refractory canine OE [3,19]. Our findings in vivo and in vitro demonstrate that this new otic solution is effective in managing canine OE, especially cases involving S. pseudintermedius and Malassezia spp. In addition, because this product is veterinarian administered, the chances of treatment failure by improper application or low compliance can be reduced. Finally, no safety related issues were reported during the study. Therefore, the combination of florfenicol, terbinafine, and mometasone furoate is highly recommendable in the treatment of canine OE.
Notes
The authors declare no conflict of interest.
Acknowledgements
This work was supported by Elanco Animal Health Korea Co., Ltd.