A comparison of traditional and quantitative analysis of acid-base and electrolyte imbalance in 87 cats
Article information
Abstract
Acid–base disorder is a common problem in veterinary emergency and critical care. Traditional methods, as well as the Stewart method based on strong ion difference concepts and the Fencl–Stewart method, can be used to analyze the underlying causes. On the other hand, there are insufficient comparative study data on these methods in cats. From 2018 to 2020, 327 acid–base analysis data were collected from 69 sick and 18 healthy cats. The three most well-known methods (traditional method, Stewart method, and Fencl–Stewart method) were used to analyze the acid–base status. The frequency of acid–base imbalances and the degree of variation according to the disease were also evaluated. In the traditional acid-base analysis, 5/69 (7.2%) cats showed a normal acid-base status, and 23.2% and 40.6% of the simple and mixed disorders, respectively. The Fencl–Stewart method showed changes in both the acidotic and alkalotic processes in 64/69 (92.8%), whereas all cats showed an abnormal status in the Fencl–Stewart method (semiquantitative approach). The frequencies of the different acid-base imbalances were identified according to the analysis method. These findings can assist in analyzing the underlying causes of acid–base imbalance and developing the appropriate treatment.
Introduction
Acid–base and electrolyte disorders are commonly found in critically ill patients in human and veterinary medicine [1-4]. In particular, metabolic acid–base disorders have been reported in various critical diseases, such as diabetic ketoacidosis, hepatitis, and kidney disease [1,2,5]. In cats, acid–base disorders have prognostic relevance, and the bicarbonate concentration in feline patients is inversely proportional to mortality [6,7]. Therefore, accurate diagnosis and proper treatment of acid–base disorders in critically ill patients are essential.
Various acid–base analysis methods, such as traditional and physicochemical approaches, have been developed to manage acid–base disorders successfully. The traditional approach called the physiologic approach is based on the Henderson–Hasselbalch equation to evaluate the acid–base status using the pH, partial pressure of carbon dioxide (pCO2), bicarbonate (HCO3−), anion gap (AG), and base excess (BE) [8]. This method can describe the compensations [9], but it has the disadvantage of being incompetent in detecting complex metabolic acid–base disorders, which are often found in severely ill patients [10,11]. Physicochemical approaches include the Stewart method (strong ion model, quantitative approach) [12-14] and the Fencl–Stewart method (semiquantitative approach) [15] and are more useful for analyzing the underlying cause than the traditional method. The strong ion difference (SID) and the total quantity of nonvolatile weak acids [AToT] can be calculated from the measured parameters to estimate the magnitude of their contribution to the BE [15] from the Stewart method. The Fencl–Stewart method calculates the effect of individual contributors (free water, chloride, albumin, phosphorus, and lactate concentrations) to estimate the magnitude of their contribution to the BE [15]. Although several studies have compared analytical methods [3,16,17], there is no consensus on which approach is superior, particularly in cats.
This study compared the interpretations of acid–base disorders using the traditional, quantitative, and Fencl–Stewart methods and analyzed the metabolic acid–base disorders in cats using the three methods. This would elucidate the pathophysiology of acid–base disorders arising from certain diseases and conduct an underlying cause analysis.
Materials and Methods
Three hundred and twenty-seven acid–base analysis data from 69 sick cats collected from 2018 to 2020 were reviewed retrospectively. Furthermore, 18 healthy client-owned cats served as the control data. The study was conducted with the approval of the Institutional Animal Care and Use Committee of Gyeongsang National University (IACUC no. GNU-210107-T0001). The data were collected from medical records, including the following: history, case signalment, physical examination, diagnostic imaging, blood analysis results (venous blood acid–base, electrolytes, lactate, and serum biochemistry profiles), and clinical diagnosis. The blood samples were collected from either the jugular or medial saphenous veins using a syringe with a 23G needle or a 24G intravenous catheter. Acid–base, electrolyte, and lactate analyses were conducted using a benchtop blood gas analyzer (pHOx Ultra Blood Gas Analyzer; Nova Biomedical, USA). Serum biochemistry analyses were conducted using a chemistry analyzer (IDEXX Catalyst One; IDEXX Laboratories, USA).
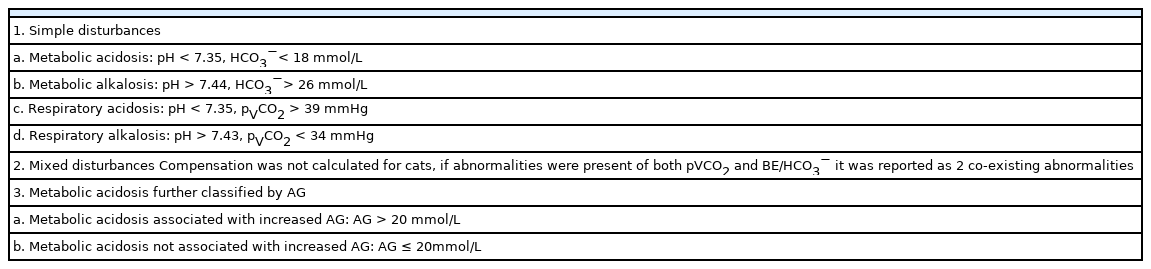
Supplementary Table S1 describes the calculated formula for the acid–base analysis [8,18,19]. The base excess of extracellular fluid (BEECF), HCO3−, and standardized base excess were calculated using pHOx Ultra Blood Gas Analyzer. Acidosis and alkalosis were defined using the pH reference based on 18 healthy cats. Table 1 lists the diagnostic criteria traditional acid–base analysis method for cats [8]. The definition of each acid–base disorder value was assumed to be ± 2 standard deviations above or below the mean of the comparison value. The statistical significance was set to p < 0.05. Statistical analyses were performed using IBM SPSS Statistics ver. 25.0 (IBM Corp., USA) and illustrated using GraphPad Prism 7 for Windows (GraphPad Software, USA). After the normality test using the Shapiro–Wilk test, all dependent variables of each acid–base analysis method between the healthy and each disorder groups using independent t-tests (parametric) or Mann-Whitney U tests (non-parametric) were compared: (pH, pCO2, HCO3−, SID, strong ion gap [SIG], ATOT, free water effect, chloride effect, phosphorus effect, albumin effect, lactate effect, unmeasured anion effect); electrolytes (Na+, K+, Cl+, Mg2+, and Ca2+); lactate; AG; and BE.
Results
Sixty-nine sick cats were analyzed: 29 neutered males, 4 intact males, 24 spayed females, 8 intact females, and 4 with no sex information. The median age was 6 years (6 months to 20 years). Most were domestic shorthair (n = 29, 42.0%), followed by mixed (n = 16, 23.2%), Russian Blue (n = 6, 8.7%), Persian (n = 4, 5.8%), Turkish Angora (n = 4, 5.8%), Abyssinian (n = 3, 4.3%), American Shorthair (n = 2, 2.9%), Munchkin (n = 2, 2.9%), Scottish Fold (n = 2, 2.9%), and Siamese cats (n = 1, 1.4%). There was no information regarding the breed of 11 cats. One or more diagnoses were recorded in 66 out of 69 cats. In contrast, 18 healthy cats included 8 neutered males (44.4%), 2 intact males (11.1%), and 8 spayed females (44.4%), with a mean age of 4 years (range, 1 to 13 years). The most common breed was the domestic shorthair (n = 14, 77.8%), while the others included Munchkin, Scottish Fold, Persian, and Turkish Angora.
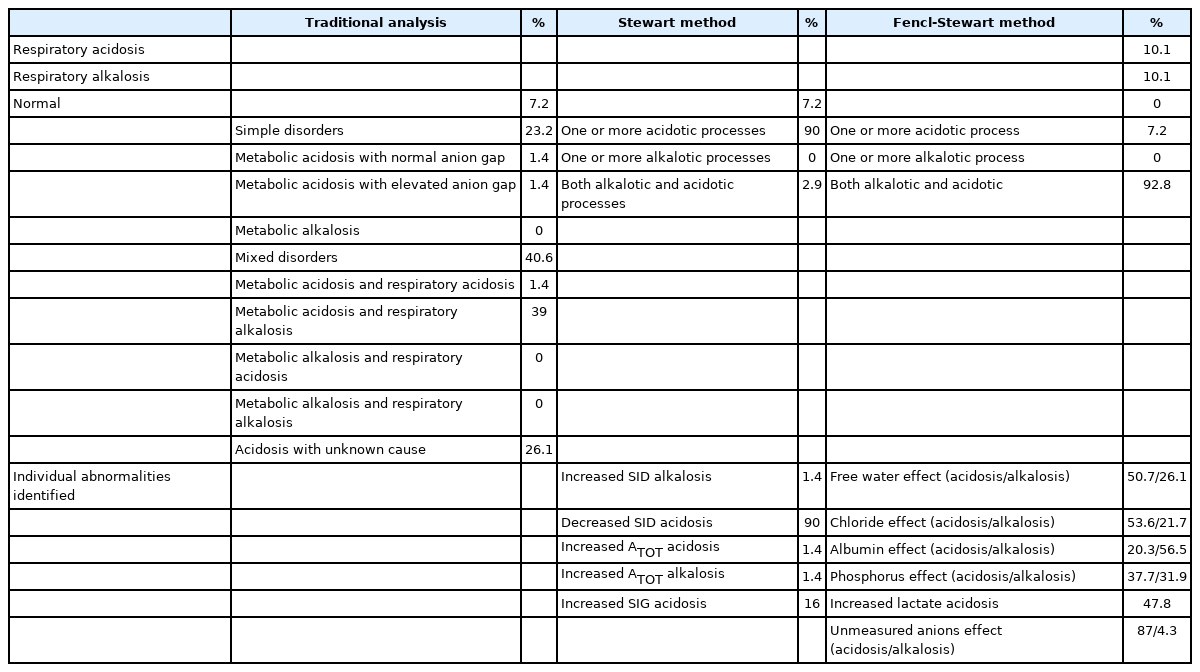
Based on the data from the 18 healthy cats, acidosis and alkalosis were defined as cases with a pH < 7.35, and > 7.44, respectively. In the traditional acid–base analysis, 5/69 cats (7.2%) showed a normal acid–base status, and 23.2% and 40.6% showed simple and mixed disorders, respectively. Acidosis could not be explained by the traditional acid–base method (unknown cause) in 18/69 patients (26.1%). Similarly, 5/69 cats (7.2%) demonstrated a normal metabolic acid–base status in the Stewart method. On the other hand, 62/69 cats (89.9%) showed one or more acidotic processes, and SID acidosis was 62/69 (89.9%). All cats showed an abnormal status in the Fencl–Stewart method (semiquantitative approach) (Table 2). Both acidotic and alkalotic processes were the most common disorders (64/69 cats, 92.8%), and one or more acidotic processes were observed in 5/69 cats (7.2%). The most common abnormality was increased unmeasured anions in 60/69 cats (87.0%), followed by decreased albumin alkalotic processes in 39/69 cats (56.5%), increased chloride-associated acidotic processes in 37/69 cats (53.6%), increased free water acidotic processes in 35/69 cats (50.7%), and increased lactate in 33/69 cats (47.8%).
The diagnosis of a normal metabolic acid–base status based on the traditional acid–base analysis method agreed with the Stewart and Fencl–Stewart methods in 5/39 and 0/39 cats, respectively. The diagnosis of an abnormal metabolic acid–base status based on the traditional acid–base analysis method was noted in 30 cats. All results were in accordance with the Stewart and Fencl–Stewart methods.
Discussion
The three different analysis methods showed different diagnoses. This was similar to a human study, in which the Stewart method was more useful for detecting metabolic acid–base disorders than traditional methods [17], and that the three methods have different abilities in analyzing acid–base disorders in animals [16]. This study revealed abnormal metabolic acid–base status in 30/69 cats, 64/69 cats, and 69/69 cats using the traditional, Stewart, and Fencl–Stewart methods, respectively. These results may be related to the good sensitivity or high overdiagnosis of physicochemical methods. The most common individual acid–base disorder was revealed as decreased SID acidosis in the Stewart method and acidosis due to the increased unmeasured anion in the Fencl–Stewart method. In a previous study, increased SIG acidosis in the Stewart method and increased unmeasured anion acidosis in the Fencl–Stewart method were most frequently identified [16]. Moreover, the formula for calculating the SID included magnesium and lactate in this study. The measurement of ionized magnesium is not used widely in veterinary practice. Therefore, more precise calculation data from the cases could be obtained. To the best of the authors’ knowledge, this is the first study to apply ionized magnesium to calculate SID in cats. A variety of formulae can be used for the Stewart-type approach for acid–base analyses. Therefore, other analytic methods, such as the constable simplified strong ion model, may have provided different results.
An analysis of acid–base disorders in 69 cats showed that respiratory alkalosis (34/69 cats) and metabolic acidosis (30/69 cats) were reported most frequently in the traditional method. According to previous reports, primary metabolic acidosis is the most common in dogs, whereas metabolic alkalosis is most common in human intensive-care unit patients [20]. In this study, respiratory alkalosis (simple or mixed) was most common. In general, the most common causes of respiratory alkalosis include hyperventilation, interpretation errors, and drugs [21]. Hyperventilation caused by activation of the peripheral chemoreceptors due to hypoxemia, central neurogenic hyperventilation caused by liver disease, hyperadrenocorticism, and overzealous mechanical ventilation might affect the result. Interpretation errors, such as falsely low pCO2 due to blood exposure to air, might also lead to an increase in oxygen saturation and a decrease in pCO2.
The most severe acidosis (pH = 7.001) was observed in a cat with a postoperative low perfusion status for tumor removal from the uterus. The most severe alkalosis (pH = 7.554) was identified in a case of iatrogenic alkalosis induced by bicarbonate administration. These are well-known and common causes of severe acid–base disorders. Metabolic acidosis caused by cancer or inadequate perfusion has been reported in human and veterinary medicine [22]. According to previous studies, lactic acid fermentation in patients with tumors causes metabolic acidosis. Moreover, the acidosis effect of cancer that affects its exacerbation, metastasis, resistance against chemotherapy, and tumor behavior, and acidosis mechanism is often set as the treatment goal [22]. Lactic acidosis is a common cause of metabolic acidosis in critically ill patients [23]. Inadequate perfusion leads to hypotension and hypoxia in these tissues, resulting in lactic acidosis. Bicarbonate administration has been reported to be a common cause of metabolic alkalosis in human and veterinary medicine [24]. Some studies have shown that the causes of metabolic alkalosis are similar between humans and animals. The causes of metabolic alkalosis in humans include bicarbonate overdose, diuretics, and citrate overdose during therapeutic plasma exchange or blood transfusion [24]. In animals, the causes of metabolic alkalosis include bicarbonate overdose, diuretics, gastrointestinal tract obstruction, and loss of gastric acid [25-28].
The reference interval of pH from 18 healthy cats was 7.39 to 7.41 in this study. In previous studies on the pH reference interval and blood gas analysis, a study of 24 cats suggested a pH reference range of 7.24 to 7.44 [29]. Another study of 8 cats suggested a pH reference range of 7.34 to 7.43 [29]. The reference ranges are essential for clinicopathological evaluations. Therefore, the difference between the reference ranges may critically affect the interpretation and diagnosis of acidosis/alkalosis. The pH reference interval identified in this study was narrow compared to the existing study results [29]. These results may lead to an increase in the rate of acidosis and alkalosis during the diagnosis of acid–base imbalances. A large sample size study (224 dogs) was conducted to evaluate the reference interval of the acid–base parameters [30]. On the other hand, studies comprising a large sample size of cats are lacking. An analysis of the acid–base parameters in 24 cats has been reported [29], and other studies have examined the blood gas acid–base parameters in conscious cats [31,32]. Another study compared a small number of cats [16]. On the other hand, there is still a lack of studies on a large sample of cats, suggesting the need for further research on cats.
In conclusion, physicochemical methods, such as the Stewart and Fencl–Stewart methods appeared to have greater diagnostic ability in metabolic acid–base disorders than the traditional approach in cats. Further prospective studies will be needed to determine the diagnostic and prognostic value of acid–base analysis in veterinary patients and determine the gold standard analysis method for cats.
Notes
The authors declare no conflict of interest.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT & Future Planning (2020R1C1C1008675).
Supplementary Materials
Supplementary data are available at https://doi.org/10.14405/kjvr.2021.61.e40.