 |
 |
| Korean J Vet Res > Volume 62(1); 2022 > Article |
|
Abstract
A 7-year-old Scottish Straight cat presented with hypersalivation and generalized hyperesthesia. The cat was provided tuna-based food for 2 to 3 weeks before clinical symptoms appeared. Abdominal ultrasonography showed a heterogeneous, hyperechoic mesentery with hypoechoic foci. Pansteatitis was tentatively diagnosed and the cat was treated with vitamin E, anti-inflammatory drugs, and dietary changes. The clinical signs and changes in mesenteric fat on serial ultrasonography resolved. After feeding tuna-based food again, the same clinical signs and mesenteric changes on ultrasound recurred, which recovered through the same treatment. This study suggests that ultrasonography can be useful for diagnosis and monitoring of feline pansteatitis.
Feline pansteatitis is a nutritional disease characterized by a marked inflammation of the fat tissue and deposition of a ceroid pigment in adipocytes, as a result of vitamin E deficiency [1,2]. If the diet contains low amounts of vitamin E, a biological antioxidant, dietary and body fat undergo oxidative degradation, leading to the formation and accumulation of reactive peroxides in cat’s adipose tissue, resulting in pansteatitis [3]. The main clinical signs of pansteatitis are fever, lethargy, inappetence, pain on skin palpation, and subcutaneous nodular masses that may show a smooth or a lumpy consistence [1]. In advanced stages of the disease, affected cats lose agility, are unwilling to move, and show hyperesthesia, especially over the back and of the abdomen [1,3]. The treatment for pansteatitis includes dietary changes, vitamin E supplementation, supportive therapy, and administration of corticosteroids [1-3]. Prognosis is known to be fair with treatment, but must be regarded as potentially poor depending on disease severity [2,3].
Diagnosis of pansteatitis could be confirmed by histological findings. On histological examination, firm, nodular fat tissues that contain acid-fast ceroid pigment, including end-products of lipid peroxidation, were observed [2]. Although it is a definitive diagnostic tool for pansteatitis, histological examination is an invasive test, thus, identifying fat degeneration noninvasively before biopsy could help in diagnosing pansteatitis. Ultrasonography has been used to identify fat changes in dogs and cats [4-7]. Ultrasonographic characteristics of feline pansteatitis have been reported in one cat as hyperechoic echogenicity of the omentum with hypoechoic foci [6]. However, the use of ultrasonography in diagnosing and monitoring feline pansteatitis has not been reported. This report describes the ultrasonographic findings in feline pansteatitis and its serial monitoring according to the treatment and shows the prognosis of rapidly diagnosed feline pansteatitis.
A 7-year-old, spayed female, Scottish Straight cat presented with hypersalivation, generalized hyperesthesia, decreased appetite, and depression. The cat had been eating homemade foods, mainly composed of chicken and duck meat; however, the cat was provided commercial canned tuna-based food (Necoya, tuna-based can, natural core) with omega-3, 2 to 3 weeks before clinical symptoms appeared. On physical examination, the cat had a fever (40.4℃) and abdominal pain. A small, irregular subcutaneous nodule was palpated at the umbilical region. A complete blood cell count was unremarkable. Increased feline serum amyloid A (fSAA, 38.4 μg/mL; reference interval, 0 to 5 μg/mL) was found at serum chemistry. Abdominal radiography depicted decreased serosal detail at the midabdomen (Fig. 1). Mottled, irregular, heterogeneous opacity of the falciform fat area and mildly increased opacity at the inguinal fat area were also detected. Abdominal ultrasonography revealed a hyperechoic and heterogeneous mesenteric fat tissue with hypoechoic nodules (Fig. 2A). The pancreas was normal in size and echogenicity, and there was no adjacent fat change. Intestinal wall layering and thickness was normal. There was no abdominal lymph node enlargement. Fat at falciform fat area showed a heterogeneously and mildly increased echogenicity. A subcutaneous nodule at the umbilical region was normal fat echogenicity and diagnosed as umbilical fat hernia. Pansteatitis was tentatively diagnosed based on dietary history, clinical signs, radiography, and ultrasonography. Medical treatment and dietary changes were prescribed during hospitalization.
The cat was treated with vitamin E (α-tocopherol, 50 IU/kg, once a day [sid]) given perorally (PO) and intravenous fluids (lactated Ringer’s solution, 15 mL/kg/h) for approximately 1 week. The cat also received amoxicillin-clavulanic acid (5 mg/kg, three times a day, intravenously; Amocla; Kuhnil Pharm Co., Ltd., Korea) and marbofloxacin (2.5 mg/kg, twice a day [bid], subcutaneously; Marbocyl 2%; Vetoquinol SA, France). Prednisolone (0.5 mg/kg, sid, PO; Solondo; Yuhan Co., Ltd., Korea) was additionally administered 3 days from the first treatment. The diet was also changed to Hill’s prescription diet i/d and z/d for 1 week. Ultrasound examination was conducted at 1, 3, and 7 days during treatment, showing a gradual decrease in the heterogeneity of the mesenteric fat (Fig. 2B-D). Marked clinical improvement involving all signs was observed within 1 week of treatment; thus, the cat was discharged on a continued course of prednisolone (0.5 mg/kg, sid, PO), amoxicillin-clavulanic acid (20 mg/kg, bid, PO), and marbofloxacin (3.5 mg/kg, bid, PO). The owner was instructed to change the cat’s diet to non-fish foods and vitamin E (30 IU) daily.
However, the owner kept feeding the same diet while giving medicine after discharge. The same clinical signs, including fever (39.4℃) and depression recurred, and increased fSAA (8.7 μg/mL) was identified at serum chemistry. Abdominal ultrasonography revealed similar mesenteric fat degeneration, including hyperechoic echogenicity of the mesenteric fat with hypoechoic nodules (Fig. 3). The cat was rehospitalized and treated for pansteatitis with the same medication and non-fish food diet. The clinical signs improved, and ultrasonographic features of the fat degeneration resolved after the treatment. At 4 months after continuing non-fish food diet and vitamin E, the cat remained in good condition with no recurrence.
Pansteatitis is uncommon in cats, and it is caused by diets containing oily fish (tuna and sardines), rich in unsaturated fatty acids relative to vitamin E [2,3,5]. Other causes, such as feeding primarily with liver, a varied diet with only small amounts of fish, pig’s brain, or commercial cat food have been reported [1,3,7,8]. In addition to nutritional factors, pansteatitis in cats is rarely described with other conditions, including abdominal injury, pancreatitis, pancreatic adenocarcinoma, intestinal lesion, and recent surgery of trauma [4,5,9]. However, in this case, the cat had no history of pancreatic disease or recent abdominal injury. The cat had clearly identified history of fish-based diet before clinical signs occurred, and clinical signs and mesenteric degeneration were improved after the dietary change. The patient was presumptively diagnosed with pansteatitis.
Ultrasonographic characteristics of feline pansteatitis have been reported as hyperechoic echogenicity of the omentum with hypoechoic foci in one cat [5]. Similarly, in this case, hyperechoic and heterogeneous echogenicity and edema of the mesenteric fat with focal hypoechoic nodules appeared on ultrasonography on the day of diagnosis. Pansteatitis was suspected by identifying fat degeneration using ultrasonography with the patient’s dietary history and increased fSAA. Ultrasonography could be useful in diagnosing pansteatitis because it is noninvasive and widely practiced at most veterinary clinics. In this report, as the treatment progressed and clinical symptoms improved, heterogeneity and echogenicity of the mesenteric fat and edema gradually resolved. These results suggest that ultrasound monitoring could help in determining the resolution or recurrence of pansteatitis in cats.
Although the results of this study suggest the usefulness of ultrasonography for diagnosis and monitoring of feline pansteatitis, hyperechoic fat changes with hypoechoic areas on ultrasonography can be identified in other diseases, which can cause steatitis and fat necrosis. Ultrasonographic findings of nodular fat necrosis have been reported as a nodule with a hypoechoic center and a hyperechoic rim at the omentum [6,7]. This pseudocystic, mineralized has been identified as a calcified necrotic mesenteric fat, also known as Bate’s body or cholestroma [7]. Hypoechoic nodules indicating fat necrosis and fat edema were identified in this case; however, calcified necrotic mesenteric fat was found in neither radiography nor ultrasound examination. In addition, peritoneal effusion has been revealed with severe omental necrosis [4-6], but peritoneal effusion was not found in this case. In humans, fat degeneration in steatitis is depicted as poorly defined increased echogenicity of the mesenteric fat, with marked mesenteric thickening [10]. Additionally, anechoic pseudocystic areas indicating fat necrosis are commonly seen in patients with fat degeneration, similar to this case [10].
Pansteatitis in cats due to fish-based foods is becoming scarcer, because most cats are fed commercial foods [1]. In the past, some cat foods were prepared with fishmeal and fish oil without additional antioxidants, which led to the development of pansteatitis. However, in this case, the patient was fed commercial canned tuna containing vitamin E. It is presumed that the amount of vitamin E or other antioxidants was insufficient to prevent pansteatitis [11]. Trends in nutrition for companion cats gained increased interest in terms of feeding alternatives of unconventional foods, homemade, and raw animal products. In addition, the practice of feeding noncommercial and conventional foods is increasing [12].
Considering the high prevalence of unconventional feeding practices, clinicians must be aware of the potential risks of nutritional disease occurrence and be taken to ensure a diagnosis.
As a limitation, there was no biopsy result of fat lesions. However, before the disease onset, fish-based diet was given, and clinical signs improved with vitamin E administration. Moreover, the same symptoms recurred when the same diet was given after the clinical signs had resolved, highly suggesting that it was pansteatitis.
In conclusion, when fever and increased fSAA appear in a cat, mainly fed with fish-based food, pansteatitis may be suspected. Ultrasound examination is meaningful for diagnosing and monitoring the patient before the occurrence of severe clinical symptoms. Therefore, ultrasonography can be used as an assistive diagnostic and monitoring tool for the treatment progress of pansteatitis, which was determined in this case, based on the history and clinical signs as well as the hyperechoic echogenicity of mesenteric fat with hypoechoic nodules.
Fig. 1.
Right (A) lateral and (B) ventrodorsal abdomen radiographs of a cat with pansteatitis. There is the lack of serosal detail at the midabdomen. Mottled, irregular, and heterogeneous fat area (arrow) is observed at the falciform fat region. Mildly increased radiopacity of the inguinal fat area (arrowhead) and the falciform fat region is also observed.

Fig. 2.
Ultrasound image of the mesentery (A) on the day of pansteatitis diagnosis and (B) 1 day, (C) 3 days, and (D) 1 week later. (A) Hyperechoic and heterogeneous mesenteric fat (arrow) with focal hypoechoic nodules (arrowheads) is seen at the time of diagnosis. These changes resolved after medical management. (B) On follow-up examination after the beginning of therapy, mesenteric echogenicity decreased (1 day later). (C) Three days after treatment, the size and heterogeneity of mesentery decreased in comparison to the first day. (D) Seven days after treatment, the cat completely recovered, with homogeneous echogenicity of the mesenteric fat.
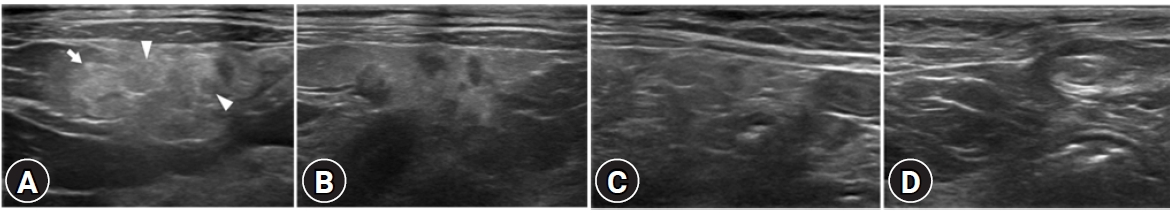
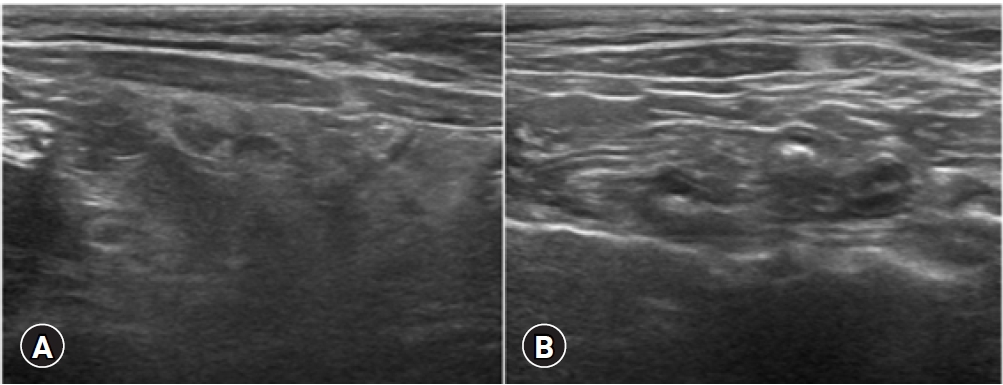
Fig. 3.
(A) Recurred pansteatitis and (B) recovery after the same treatment. (A) Hyperechoic echogenicity of the mesenteric fat with a mild hypoechoic nodule (arrow) was detected at the time of pansteatitis recurrence. (B) The recheck ultrasound examination of the mesenteric fat showed normally hypoechoic and homogeneous echogenicity of the mesenteric fat.
References
1. Niza MM, Vilela CL, Ferreira LM. Feline pansteatitis revisited: hazards of unbalanced home-made diets. J Feline Med Surg 2003;5:271-277.

2. Koutinas AF, Miller Jr WH, Kritsepi M, Lekkas S. Pansteatitis (steatitis, “yellow fat disease”) in the cat: a review article and report of four spontaneous cases. Vet Dermatol 1992;3:101-106.



3. Tidholm A, Karlsson I, Wallius B. Feline pansteatitis: a report of five cases. Acta Vet Scand 1996;37:213-217.
4. Hecht S, Henry G. Sonographic evaluation of the normal and abnormal pancreas. Clin Tech Small Anim Pract 2007;22:115-121.


5. Zini E, Hauser B, Ossent P, Dennler R, Glaus TM. Pansteatitis and severe hypocalcaemia in a cat. J Feline Med Surg 2007;9:168-171.


6. Griffin S. Feline abdominal ultrasonography: what’s normal? what’s abnormal?: abdominal lymph nodes, peritoneal cavity and aorta. J Feline Med Surg 2021;23:835-849.


7. Schwarz T, Morandi F, Gnudi G, Wisner E, Paterson C, Sullivan M, Johnston P. Nodular fat necrosis in the feline and canine abdomen. Vet Radiol Ultrasound 2000;41:335-339.


9. Fabbrini F, Anfray P, Viacava P, Gregori M, Abramo F. Feline cutaneous and visceral necrotizing panniculitis and steatitis associated with a pancreatic tumour. Vet Dermatol 2005;16:413-419.


- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,723 View
- 87 Download
- ORCID iDs
-
Hyesun Kang

https://orcid.org/0000-0001-6221-4176Daji Noh

https://orcid.org/0000-0002-1447-2194Sang-Kwon Lee

https://orcid.org/0000-0002-3097-0345 - Related articles


 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



