Peritoneal lymphomatosis with pleural and peritoneal effusions in a dog: a case report
Article information
Abstract
Peritoneal lymphomatosis (PL) is a rare lymphoid neoplasm in dogs. A nine-year-old spayed female Labrador retriever presented with pleural and peritoneal effusions. Diagnostic imaging revealed diffuse nodular to massive lesions in the mesentery, particularly in the caudal abdomen. While the superficial lymph nodes did not show significant changes, enlargement was observed in the intra-abdominal and intra-thoracic lymph nodes. Cytological and flow cytometric analyses of the effusion indicated the presence of large B-cell lymphocytes expressing CD3-/CD5-/CD14-/CD21-/CD34+/CD45+/CD79a+. PL was diagnosed using diagnostic imaging and fluid analysis. This case report highlights the clinical and diagnostic features of canine PL.
Peritoneal tumors typically develop as secondary growths. The peritoneal surface can be affected by three types of cell lines in a secondary manner: epithelial neoplasia causing carcinomatosis, mesenchymal neoplasia leading to sarcomatosis, and lymphoid malignancy resulting in lymphomatosis. Peritoneal lymphomatosis (PL) refers to the spread of lymphoma cells throughout the peritoneum and abdominal organs, causing widespread involvement of the peritoneal cavity [1].
PL has rarely been reported in dogs. Due to the limited understanding of its underlying causes, information regarding the diagnosis, treatment, and prognosis of lymphomatosis is scarce [2]. The precise mechanism by which lymphoma affects the peritoneum remains unknown because the omentum lacks lymphoid aggregates in dogs [3]. This report describes the clinical manifestations and diagnostic characteristics of canine PL.
A 9-year-old spayed female Labrador retriever presented with ascites and chronic diarrhea. The dog had been previously treated with prednisolone, mycophenolate mofetil, and antibiotics for a month at a local animal hospital considering the possibility of protein losing enteropathy without further blood or ascites examination. The drug displayed no significant improvement.
Physical examination revealed depressed behavior, a low body condition score of two out of nine, tachypnea, tachycardia, and hypotension. Auscultation revealed muffled heart sounds, which were subsequently confirmed to be attributable to pleural effusion. The superficial lymph nodes showed no remarkable changes, but a hard mass was palpable in the lower abdominal cavity, located anterior to the bladder.
Body cavity fluid analysis was performed after ultrasonography-guided thoracocentesis and paracentesis. The peritoneal and pleural effusions appeared yellowish, and cytological analysis confirmed the presence of exudates. Cytologic examination of the fluid samples (Fig. 1A) showed that over 50% of the cells were round with a high nuclear-to-cytoplasmic (N:C) ratio. The nuclei were more than twice the size of red blood cells, with prominent 1 to 2 nucleoli and irregular margins in the nuclei and cytoplasm. Abnormal mitotic figures and karyorrhexis were occasionally observed. Approximately 30% of the cells in the peritoneal effusion were neutrophils. No evident degenerative changes, phagocytosis, or infectious agents were identified. In the pleural effusion, 40% of the cells were macrophages, and no intracytoplasmic phagocytosis or multinucleated cells were observed. No infectious agents were detected in the pleural effusion. Subsequent cultures of the pleural and peritoneal fluids did not show any growth. Based on the cytological findings, neoplasia originating from round cells was suspected, with high-grade lymphoma considered the most likely differential diagnosis. Peripheral blood analysis showed no significant findings, except for neutrophilia with left shift (segmented neutrophil: 29.39 × 109/L, band neutrophils: 2,942 cells/μL) and hypoalbuminemia (2.1 g/dL). Flow cytometry and clonality tests were used to verify the immunophenotype of malignant round cells, as described previously [4].

Cytology of the cytospin preparation for pleural effusion (A) and fine-needle aspiration (FNA) of the sternal lymph node (B). (A) Malignant round cells have scant to moderate cytoplasm, prominent nucleoli, nuclei more than twice the size of blood cells, and occasionally an abnormal mitotic figure (Diff-Quik staining, scale bar: 100 μm). (B) FNA cytology of the sternal lymph nodes. Prominent epithelioid histiocyte infiltration is observed (Diff-Quik staining, scale bar: 100 μm).
Flow cytometric analysis of the neoplastic cells demonstrated CD3-/CD5-/CD14-/CD21-/CD34+/CD45+/CD79a+, which showed the presence of immature large B-cell lymphocytes (Fig. 2). Polymerase chain reaction tests of pleural and peritoneal fluids for antigen receptor rearrangement (PARR) indicated polyclonal results for both the immunoglobulin and T-cell receptor genes (IDEXX Laboratories, USA).
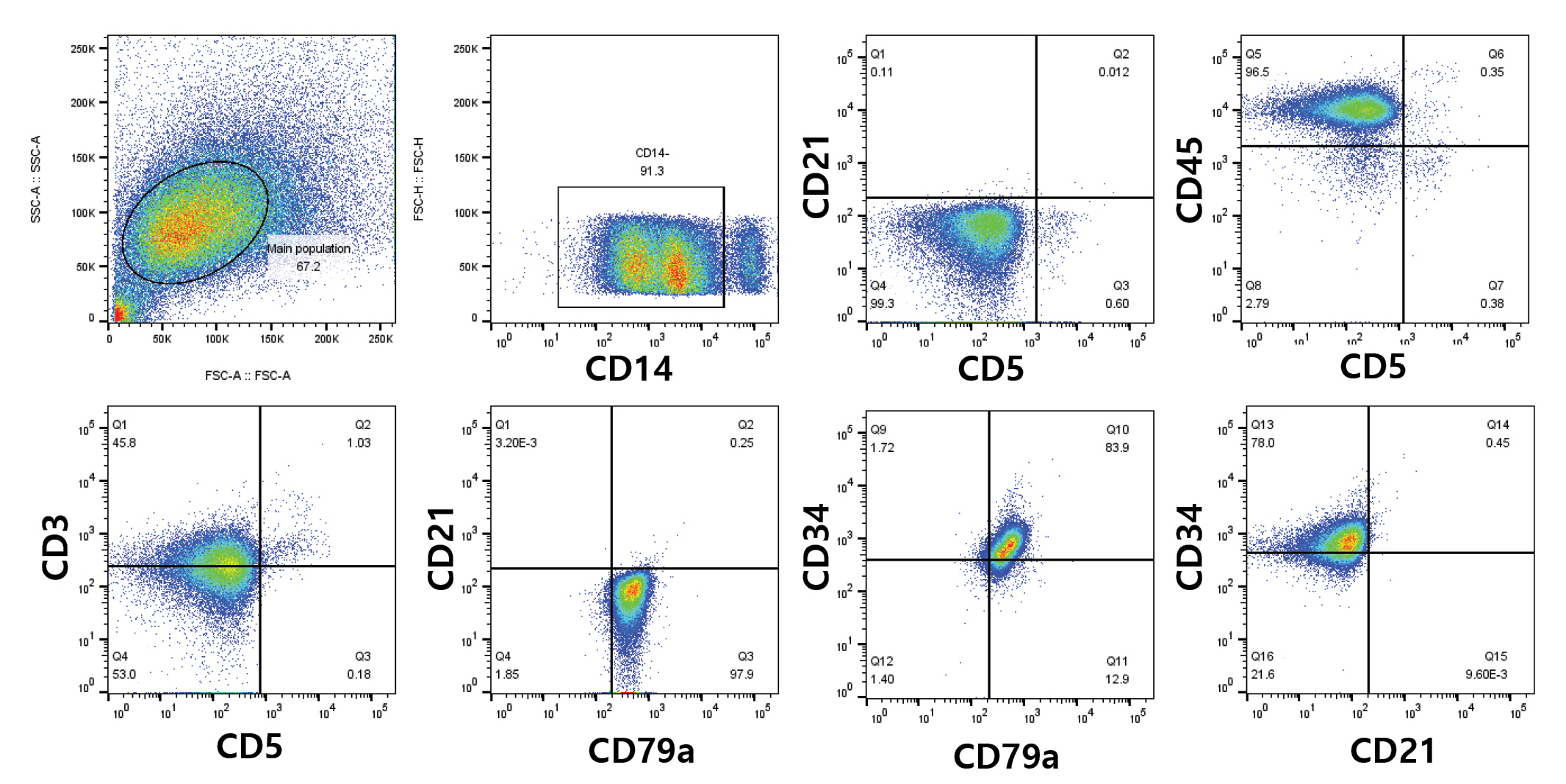
Immunophenotyping assay by flow cytometric analysis of the pleural effusion. Lymphocytes predominantly showed CD3-/CD5-/CD14-/CD21-/CD34+/CD45+/CD79a+.
To determine the origin of the neoplasia, ultrasonography and non-anesthetized computed tomography (CT) were performed. The mesentery throughout the abdominal cavity exhibited nodular to massive lesions, particularly at the level of the caudal abdomen (Fig. 3). Enlargement of the intra-abdominal, sternal, mediastinal, and tracheobronchial lymph nodes was observed, along with significant pleural and peritoneal effusions. Ultrasonography revealed severe thickening of the gastric wall at the level of the pylorus, loss of wall layers, and heterogeneous echogenicity. The duodenal wall exhibited thickening, partial loss of layering, and corrugation with hyperechoic coalescing nodules identified in the adjacent mesenteric fat. A hypoechoic nodule was detected in the right hepatic lobe.
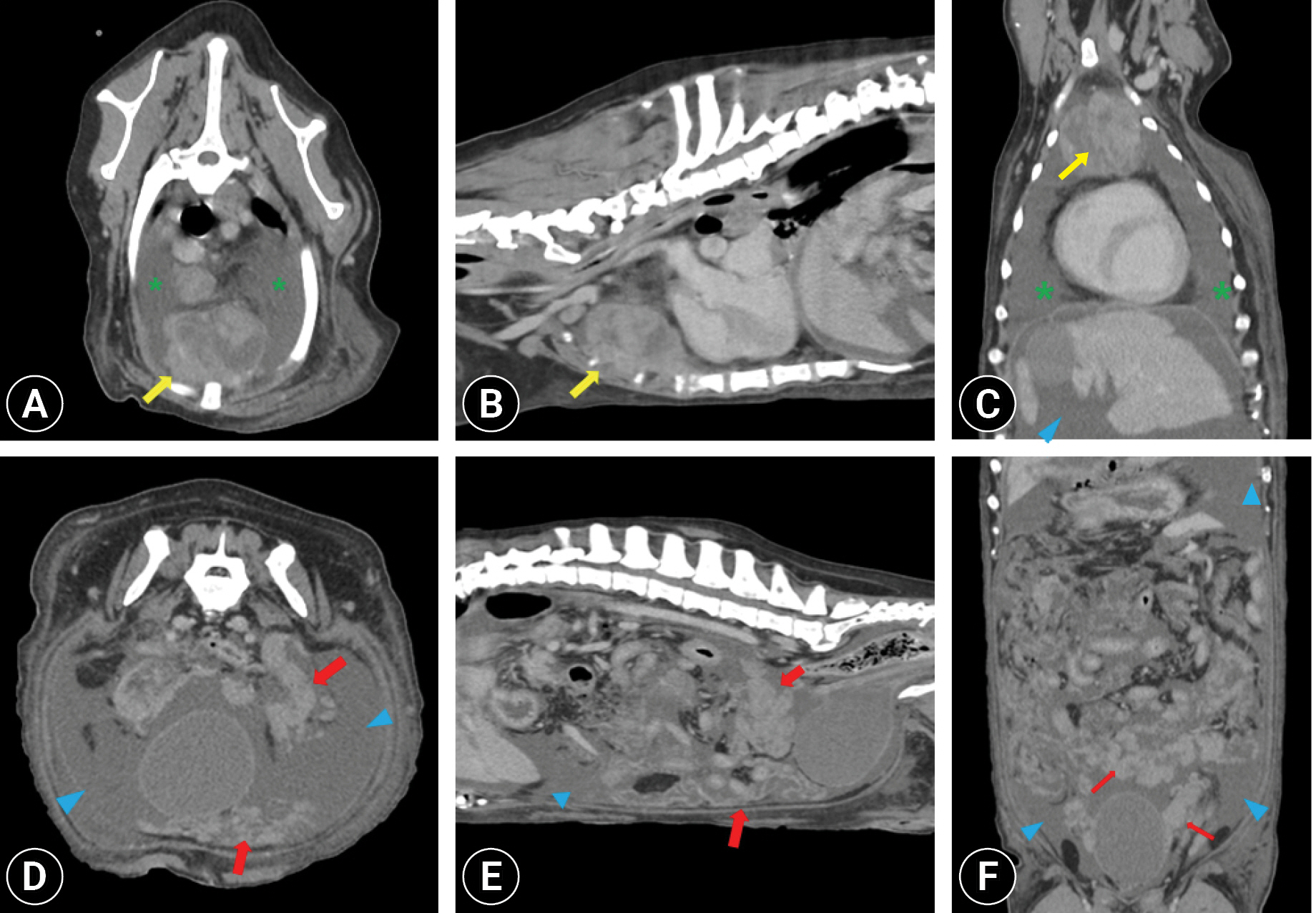
Non-anesthetized thoracic and abdomen computed tomography images. Post-contrast (A, D) transverse, (B, E) sagittal, and (C, F) dorsal plan images showing disseminated nodular to massive infiltration around the mesentery (red arrows), especially anterior to the urinary bladder with peritoneal effusion (blue arrowheads). Massive enlargement of the sternal lymph node (yellow arrows) and pleural effusion (green asterisks) were identified.
Based on the cytological and flow cytometric results indicative of large B-cell lymphoma, along with the imaging characteristics of the massive mesenteric lesion, PL was the most likely differential diagnosis.
However, ultrasound-guided fine-needle aspiration (FNA) of the mesenteric mass and sternal lymph nodes revealed low cellularity characterized by a few round cells and marked infiltration of epithelioid histiocyte-like cells (Fig. 1B). Histiocyte-like cells were observed either in clusters or individually and exhibited epithelioid, fusiform, or polygonal shapes. These cells exhibited moderate anisokaryosis, distinct nucleoli, and coarse chromatin. Microscopic images revealed multinucleated cells with multiple cytoplasmic vacuoles and intracytoplasmic phagosomes. Flow cytometric analysis was not performed because of the low cellularity. Therefore, the possibility of histiocyte-derived neoplasia or granulomatous inflammation cannot be disregarded.
Following the owner’s request, the dog was discharged from the hospital. While the owner was deliberating the possibility of surgery or other medical interventions, the dog continued to exhibit severe anorexia and recurrent ascites. Despite the intention to confirm the diagnosis through histopathology using a surgical or true-cut biopsy, the dog died at home on the fourth day after the initial presentation. No postmortem examination was performed.
PL, a rare condition in dogs, has previously been reported in only one case, which involved a dog with large granular lymphocyte intestinal lymphoma [5]. PL is characterized by the spread of lymphoma to the peritoneum. It has been suggested that neoplasms originating from epithelial cells can directly invade the peritoneum, particularly in cases of gastrointestinal and urogenital malignancies [1]. Tumor cells can detach from the primary tumor and float within the peritoneal fluid, leading to their adherence and invasion of the peritoneal surface [5]. Previous reports have described intraperitoneal lesions following tumor resection in dogs and FNA of primary masses in feline colonic lymphoma [2,6]. Tumor cells can disseminate directly from the primary tumor or through iatrogenic processes, potentially resulting in intraperitoneal seeding. Given the repeated paracentesis and ultrasound findings of multiple intra-abdominal organ and lymph node involvement, a speculative hypothesis arises regarding the possible route of peritoneal dissemination. However, the exact mechanism remains unclear without histopathological and postmortem examinations.
The misdiagnosis of peritoneal involvement in metastatic carcinoma is common in dogs with lymphomatosis. CT can provide valuable information for the diagnosis of PL. Key features include omental caking with a bulky homogeneous mass or homogeneous thickening of the peritoneum and mesentery. Other characteristic features include diffuse enlargement of the abdominal lymph and lymphomatous involvement of other intra-abdominal organs such as the liver and intestinal tract. Carcinomatosis typically presents as a small nodular pattern with affected regional lymph nodes near the primary mass [7]. Ascites are frequently observed in PL and carcinomatosis [7]. PL can manifest as an initial presentation of lymphoma during the disease course or as a transformation from an indolent subtype of lymphoma to a high-grade form [7]. In the present case, diffuse and homogeneous nodular to massive lesions were identified in the abdominal mesentery and peritoneum, along with multiple lymphadenopathies and lesions suggestive of liver and alimentary tract involvement within the abdominal cavity.
Flow cytometry, in conjunction with diagnostic imaging, is a reliable method for identifying malignant cells in body cavity effusions. Immunophenotyping of neoplastic cells revealed CD3-/CD5-/CD14-/CD21-/CD34+/CD45+/CD79a+, indicating large B-cell lymphoid neoplasia [8].
PARR demonstrated a polyclonal pattern for both the immunoglobulin and T-cell receptor genes. Compared to flow cytometry, PARR exhibits lower sensitivity (67%) and a low negative predictive value (52%) for diagnosing B-cell lymphoma, although its specificity is maintained at 100% [9]. False-negative PARR results may be attributed to tumor cells possessing genes within the V or J regions that are not amenable to primer binding, thereby hindering accurate detection [10]. Another plausible explanation is that malignant cells may consist of early precursor cells in which the gene rearrangement process may not have been completed. In this case, a significant proportion of the cells were large, round cells expressing CD34, a precursor cell marker. CD34 positivity has been reported in approximately 10% of B-cell lymphomas [11]. CD34+ B-cell lymphoma is presumed to be a distinct lymphoproliferative disorder from CD34+ leukemia and is considered an aberrant manifestation [11,12]. A combined approach involving cytopathology and flow cytometry can serve as a non-invasive and highly useful method for evaluating body cavity effusion with lymphomatous involvement [13].
The cytological findings in the pleural fluid and ascites were consistent with those of high-grade lymphoma. Cytological findings from the sternal lymph nodes and mesenteric mass showed multiple aggregates of epithelioid histiocyte-like cells, predominantly observed over round cells in the sternal lymph nodes. The cause of granulomatous reactions in tumors, including lymphomas, remains unknown; however, several possibilities have been proposed. For instance, a T-cell-mediated nonspecific inflammatory response and delayed hypersensitivity to cytokines or antigens produced by tumor cells can stimulate monocyte activation and transformation into epithelioid histiocytes [14]. Diagnosis can be challenging in cases with a prominent granulomatous lesion, as the lesion may obscure underlying malignant diseases. Distinguishing reactive histiocytosis or histiocytic neoplasia from the underlying tumor based solely on cytological findings can be difficult, and biopsy may be required [15].
This case report highlighted a rare condition known as PL in dogs that is characterized by intra-abdominal and thoracic lymphadenopathy as well as peritoneal and pleural effusion. The diagnostic approaches employed in this study, including diagnostic imaging, cytology, and flow cytometry immunophenotyping of body cavity effusion, collectively contributed to the tentative diagnosis of CD34+ large B-cell lymphoma with mesenteric involvement.
Notes
The authors declare no conflict of interest.
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2020R1C1C1008675).
