 |
 |
| Korean J Vet Res > Volume 64(1); 2024 > Article |
|
Abstract
Notes
Funding
This work was supported financially by a grant (B-1543083-2023-24-01, Animal and Plant quarantine Agency, Republic of Korea).
Author’s Contributions
Conceptualization: Yang DK; Data curation: Cheong YJ, Yang DK; Formal analysis: Yang DK; Funding acquisition: Hyun BH, Yang DK; Investigation: Yang DK; Methodology: Cheong YJ, Hyeon LS, Kim M; Project administration: Yang DK; Software: Yang DK, Kim HH; Validation: Kim HH, Lee HJ, Cheong YJ; Writing-original draft: Yang DK; Writing-review & editing: all authors.
Fig. 1.
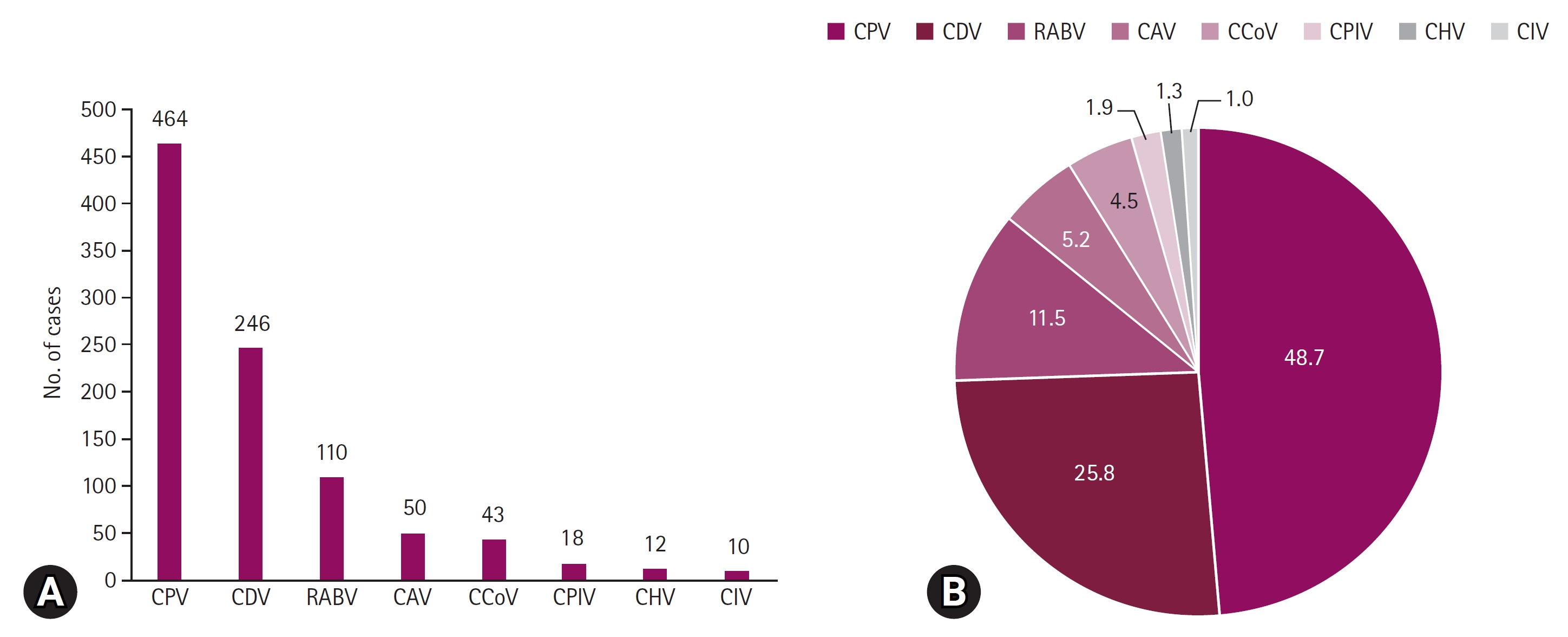
Fig. 2.
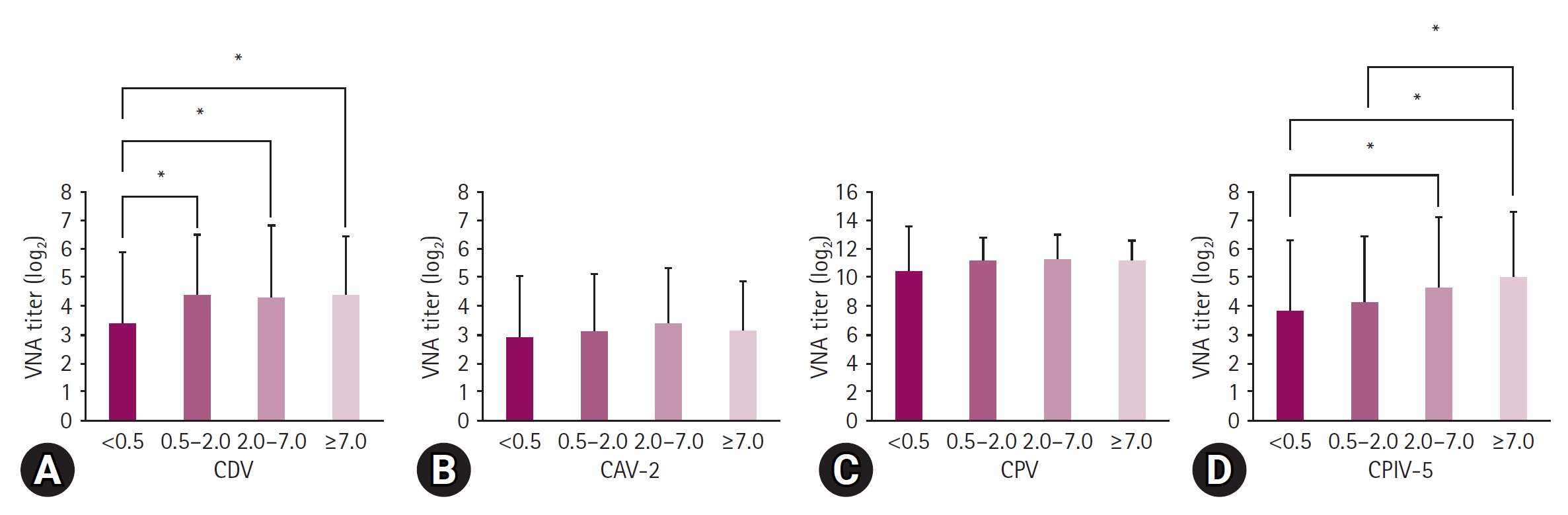
Fig. 3.
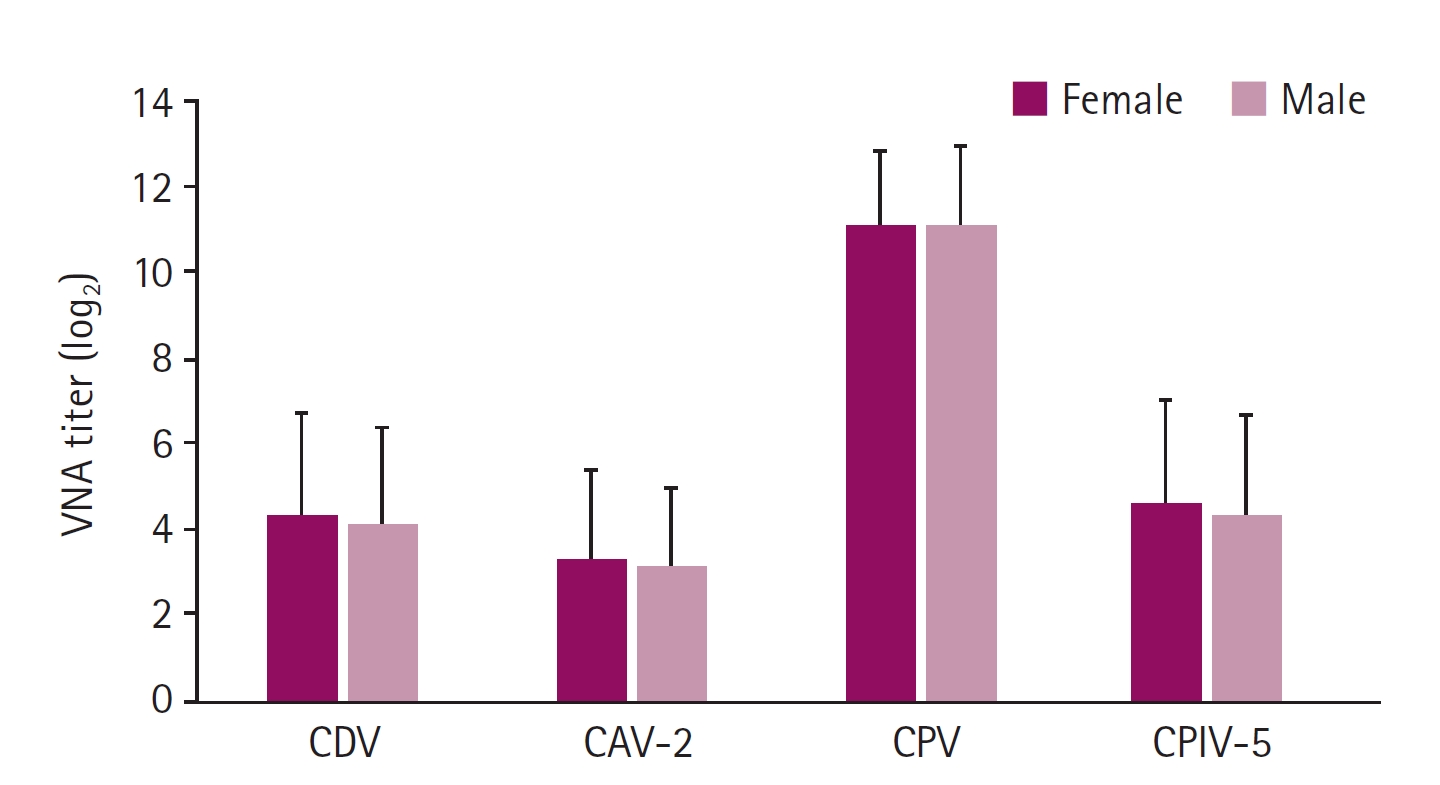
Fig. 4.
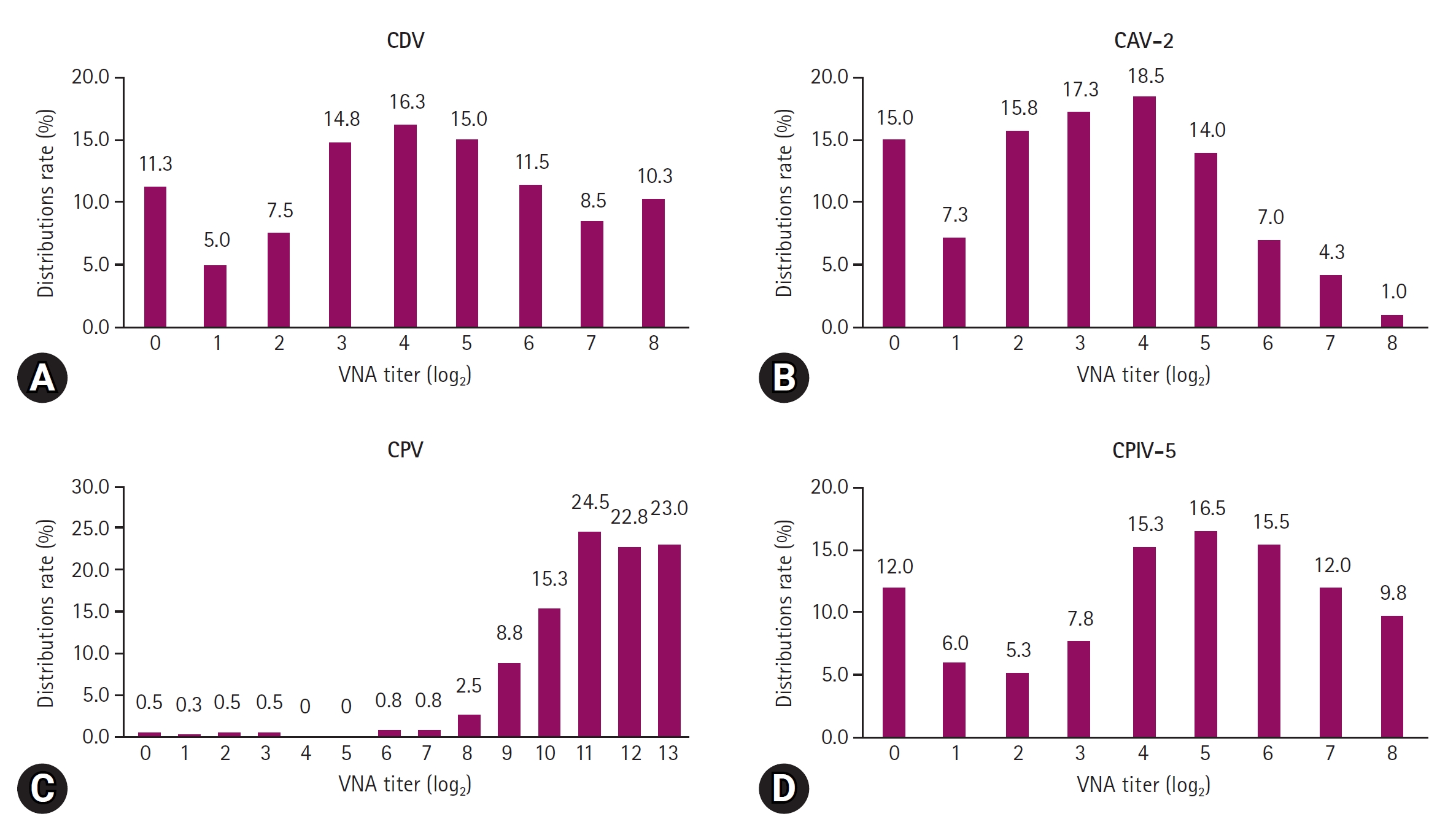
Table 1.
Table 2.
| Protective rate (%) | p-value | CPV or CIPV-5 | Protective rate (%) | p-value | |
|---|---|---|---|---|---|
| CDV | |||||
| Total | 181/400 (45.3) | Total | 393/400 (98.3) | ||
| < 0.5 | 18/50 (36.0) | 0.40 | < 0.5 | 46/50 (92.0) | < 0.01* |
| 0.5-2.0 | 65/130 (50.0) | 0.5-2.0 | 127/130 (97.7) | ||
| 2.0-7.0 | 70/157 (44.6) | 2.0-7.0 | 157/157 (100) | ||
| ≥ 7 | 28/63 (44.4) | ≥ 7 | 63/63 (100) | ||
| CAV-2 | |||||
| Total | 179/400 (44.8) | Total | 215/400 (53.8) | ||
| < 0.5 | 20/50 (40.0) | 0.51 | < 0.5 | 19/50 (38.0) | < 0.05 |
| 0.5-2.0 | 57/130 (43.9) | 0.5-2.0 | 63/130 (48.5) | ||
| 2.0-7.0 | 77/157 (49.0) | 2.0-7.0 | 92/157 (60.5) | ||
| ≥ 7 | 25/63 (39.7) | ≥ 7 | 41/63 (65.1) |
References
- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 1,982 View
- 56 Download
- ORCID iDs
-
Dong-Kun Yang

https://orcid.org/0000-0001-5765-3043Ha-Hyun Kim

https://orcid.org/0000-0001-6473-0035Hye Jeong Lee

https://orcid.org/0000-0003-2044-6176Young-Ju Cheong

https://orcid.org/0000-0002-2344-7329Lee-Sang Hyun

https://orcid.org/0000-0002-7608-156XMinuk Kim

https://orcid.org/0009-0006-5626-1420Bang-Hun Hyun

https://orcid.org/0000-0002-3429-3425 - Related articles


 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



