Lymphomatosis, the intraperitoneal dissemination of lymphoma, has scarcely been reported, despite lymphoma being one of the most common neoplasms in cats [
1,
2]. In human medicine, however, spreading routes to the peritoneum of lymphoma have been recognized. As a direct spread, lymphatic, hematogenous, and intraperitoneal seeding is observed, even though the peritoneum is comprised of fibrofatty tissue devoid of lymphoid tissue [
3,
4]. Lymphomatosis in the peritoneum has rarely been reported as secondary neoplasia or metastatic manifestations, compared to primary neoplasia from the peritoneum, despite the high incidence of feline lymphoma [
5].
Peritoneal lymphomatosis has only been reported in 8 cats, all of which had the alimentary form of lymphoma [
5-
8]. The alimentary form is the most common anatomical type of lymphoma in cats that accounts for 55% of all intestinal malignancies [
1,
2]. Most of these reports described the sonographic features of lymphomatosis [
5-
7]. There is a lack of specific diagnosis, treatment, and prognosis assessment for lymphomatosis in cats because of the insufficient number of cases. This study examined the radiography, ultrasonography, and computed tomography (CT) imaging features of alimentary lymphoma with peritoneal lymphomatosis in a cat.
A 12-year-old, 5.6 kg, castrated male Persian cat presented with abdominal distension, anorexia, and no defecation for 3 days. The physical examination revealed abdominal distension and tachypnea. The other vital signs, including temperature and pulse, were normal. The complete blood count and serum biochemistry showed an elevated total white blood cell count (22.4 ├Ś 109/L; reference, 6.4-19.6 ├Ś 109/L).
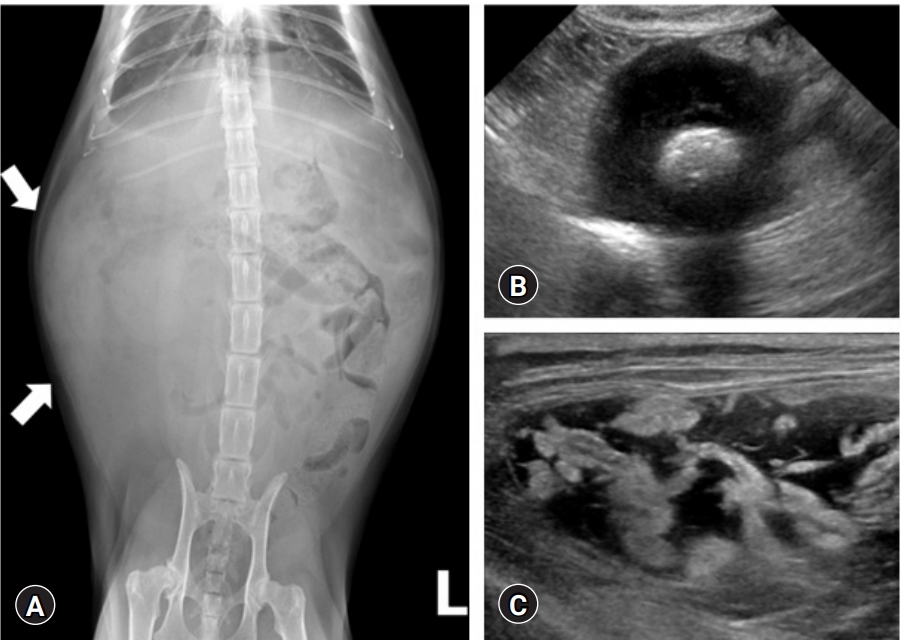
The abdominal and thoracic radiographs were obtained using digital radiographic equipment (XPLRER-900; Medien International Co., Ltd., Korea). Abdominal radiography revealed an ill-defined mass with soft tissue opacity in the left cranial abdomen. The loss of abdominal serosal detail was attributed to moderate ascites (
Fig. 1A). Thoracic radiographs exhibited pleural effusion with a cranioventral mediastinal mass.
Abdominal ultrasonography (Philips iU22 SonoCT System; Philips Ultrasound, USA) revealed the abdominal mass as a well-defined hyperechoic lesion within a thickened ileal segment. The ileal wall showed transmural loss of layering, and its thickness was approximately 17 mm (
Fig. 1B). The peritoneum showed increased echogenicity with extensive thickening. Multiple hyperechoic and hypoechoic nodules throughout the peritoneum were accompanied by mild ascites with echogenic sludge (
Fig. 1C).
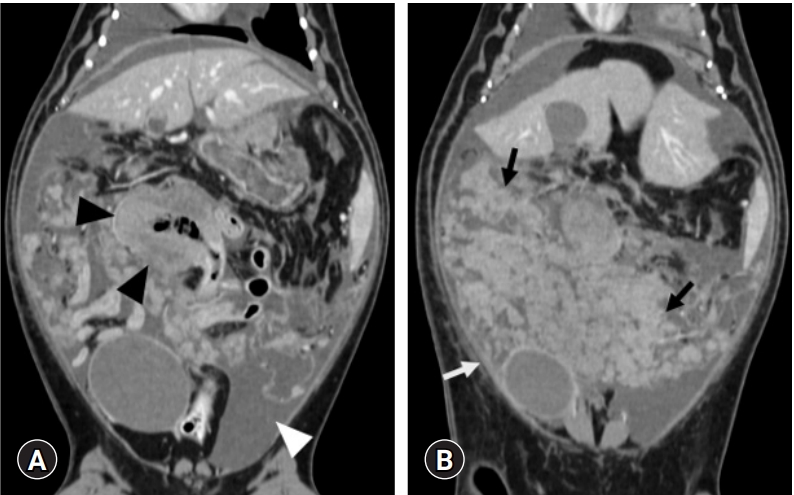
A survey CT scan using a 32-detector-row CT scanner (Alexion; Toshiba, Japan) and the following parameters (120 kVp, 150 mAs, 2 mm slice thickness, 0.75 second rotation time, and 0.938 collimation beam pitch) was performed under general anesthesia. An iodinated contrast medium (Omnipaque; GE Healthcare Ireland, Ireland) was administered into the cephalic vein at a dosage of 600 mg iodine/kg using an angiographic power injector (Salient; Imaxeon Pty. Ltd., Australia) at a rate of 1.2 mL/s. Abdominal CT revealed a bulky heterogeneous ileal mass (4 ├Ś 5 ├Ś 3 cm) with focal contrast enhancement in the artery (160 Hounsfield unit [HU]), portal (180 HU), and delayed (130 HU) phases. Diffuse discrete nodules with mesenteric involvement and peritoneal thickening with contrast enhancement in the artery (85 HU), portal (95 HU), and delayed (110 HU) phase were also recognized (
Fig. 2). On thoracic CT, enlarged bilateral sternal lymph nodes were observed with strong contrast enhancement.
Ultrasonography guided fine needle aspirations of an ileal mass and bilateral enlarged sternal lymph nodes were performed. Both showed medium to large multiple lymphoblasts with round to irregular nuclei, fine to coarse chromatin patterns, and prominent nucleoli. These lymphoblasts comprised more than 50% of the total cells. These findings indicated a high-grade lymphoma. Thoracocentesis and abdominocentesis showed modified transudate, and the direct microscopic smear detection of ascitic fluid revealed multiple lymphocytes (
Fig. 3). The cytologic evaluation suggested that the intestinal mass is a high-grade alimentary lymphoma and peritoneal lymphomatosis.
One week after clinical presentation, the patient was started on a CHOP (cyclophosphamide, hydroxydaunorubicin, vincristine, and prednisone) chemotherapy regime. A follow-up abdominal ultrasound examination was performed after the first week of the CHOP protocol (vincristine, 0.6 mg/m2) and showed no remarkable changes. After the second week (cyclophosphamide, 200 mg/m2), however, the patient showed anorexia consistently and died 2 weeks after the initial diagnosis. The owner rejected a postmortem examination.
This case report describes the radiography, ultrasonography, and CT imaging features of peritoneal lymphomatosis from an alimentary lymphoma in a cat. The specific findings, in this case, included thickened hyperechoic mesentery with multiple nodules in ultrasonography and contrast-enhanced thickened peritoneum with diffuse ill-defined nodules on the CT images. These findings could assist in the diagnosis and treatment of lymphomatosis by alimentary lymphoma.
Alimentary lymphoma is the most common (55%) of all intestinal neoplasms, followed by adenocarcinoma (32%) in cats [
1]. Another report of 14 cases of feline secondary peritoneal malignancies described lymphomatosis in 2 cats and carcinomatosis in 11 cats [
6]. The treatment of alimentary adenocarcinoma involves surgical excision, but the average survival time is one year, even in the presence of carcinomatosis [
9]. On the other hand, alimentary lymphoma can be treated with chemotherapy, and its prognosis is generally guarded [
1]. Therefore, early differentiation of the alimentary form of lymphoma from adenocarcinoma is essential for determining the treatment and prognosis considering its non-surgical and surgical treatment.
Previous studies reported the ultrasonography findings of peritoneal lymphomatosis with alimentary lymphoma in 7 cats [
5-
7]. Transmural thickening of the peritoneum with increased echogenicity was observed. In addition, multiple small hypoechoic or hyperechoic nodules from the peritoneal surfaces were also noted with mild ascites. These ultrasonography findings are consistent with the present case. Ultrasonography has been used as a more common modality for diagnosing the neoplastic, inflammatory, and infectious peritoneal pathologies in veterinary medicine, whereas CT is regarded as the gold standard for investigating abdominal pathologies in human medicine [
10,
11]. The most common CT features of lymphomatous involvement of peritoneum in human medicine are bulky homogeneous mass with gastrointestinal wall thickening, peritoneal involvement with thickening and contrast enhancement, mild to moderate ascites, retroperitoneal lymphadenopathy, and lymphomatous infiltration of other solid organs, such as liver and spleen [
11,
12]. In addition, several studies have identified the different CT appearances between 3 types of peritoneal tumor invasion: lymphomatosis, carcinomatosis, and sarcomatosis in human medicine [
3,
13,
14]. They share similar features, such as peritoneal lesions, lymphadenopathy, and ascites. On the other hand, lymphomatosis is characterized by bulky homogeneous masses, small to moderate amounts of ascites, diffuse lymphadenopathy compared to other malignancies, and is associated with solid organ involvement, particularly the spleen and liver [
12-
14]. In the case of carcinomatosis, relatively small nodules invaded the omental fat, a large amount of ascites, and no specific lymphadenopathy without surroundings of primary mass were identified [
13,
14]. Although peritoneal sarcomatosis also has bulky masses, they usually have an excessive number of blood vessels and heterogeneous parenchyma compared to lymphomatosis [
14]. In contrast to the reports of human medicine, the ileal mass in the present case was relatively small and showed heterogeneously enhanced parenchyma without retroperitoneal lymphadenopathy. The amount of ascites was similar to previous reports in human medicine. The typical CT findings described in feline peritoneal and pleural sarcomatosis, which included well-marginated, variable nodule size with heterogeneous soft tissue attenuation, remarkable rim-like enhancement (24 HU pre-contrast, 80 HU post-contrast), and cavitated, hyperattenuating mineralized foci, were not consistent with this patient [
15].
The lack of histopathological analysis was a limitation in this case. Because this study included only one cat, further studies will be needed to evaluate the CT characteristics of peritoneal lymphomatosis from feline alimentary lymphoma and the differences between other secondary peritoneal malignancies in larger populations.
This study described the CT features of peritoneal lymphomatosis originating from an alimentary lymphoma in a cat. These findings are essential for radiologists in assessing peritoneal infiltration of gastrointestinal lymphoma and helpful for selecting treatment options and evaluating the prognosis.










 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



